Review Oriented Development (ROD)
0.1 Review Gap
With powerful LLMs, code and document generation is instant; however, the bottleneck has shifted to the review phase.
Without a structured review framework, the speed gained by AI is lost in manual, error-prone verification cycles.
This is critical in Biopharma, where the cost of error is not just a bug, but a regulatory finding or patient safety risk. We cannot afford “hallucinations” in clinical data.
1 Shifting to ROD
Generative AI has fundamentally inverted the software development lifecycle. Developers are no longer strictly writers of code; they are now auditors of AI-generated content. Existing tools and processes are optimized for writing, not reviewing.
For an organization with multiple candidate compounds and clinical studies in different phases, a review oriented, AI powered, human-in-loop process built on top of propritory and open source tools will be essential to deliver A&R results that is reproducible and compliant.
2 Solutions
Implement a hierarchical YAML-based Review-Oriented Development (ROD) framework within the Biometrics team powered by LLMs.
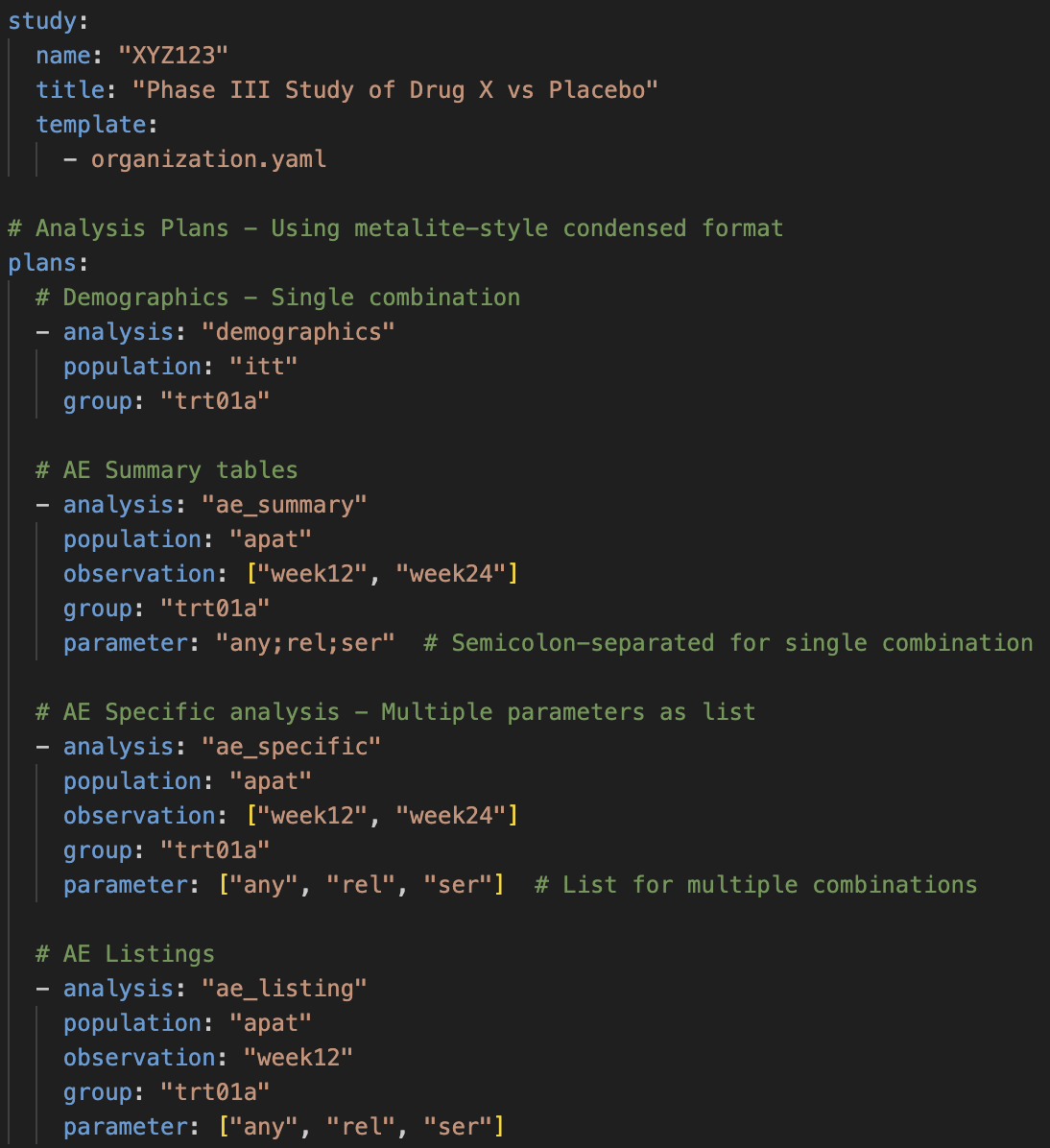
3 Key Business Benefits
| Benefit Category | Impact |
|---|---|
| Efficiency | Accelerated Time-to-Insight: Reduces review cycles by focusing on high-level logic rather than low-level syntax. |
| Compliance | Audit-Ready by Default: YAML files serve as a self-documenting history of exactly what analysis was requested and performed. |
| Quality | Enhanced Integrity: Removes the variability of code styles. If the configuration is correct, the output is guaranteed to match the standard. |
| Scalability | AI-Native Workflow: Prepares the organization to scale AI adoption safely by having guardrails firmly in place. |